 Within LUMO eigenvalues, the greatest decrease was observed for Al-4N3 (0.027 au with B3LYP/DZP). This video shows you how to draw the different molecular orbitals for a conjugated system, how to make sense of the logic without the crazy math, and how to quickly and easily identify HOMO and LUMO molecular orbitals. In the comparison of HOMOs and LUMOs energy gap, it was found that the greatest increase on HOMO eigenvalue (0.03 au at the B3LYP/DZP level) was obtained for Al-5N3. Video 2: HOMO and LUMO for Conjugated Systems This video provides you with a logic-based overview of MO theory with simple take-aways to focus on at the organic chemistry level. chance of false negative covid test at home potassium bromide for dogs dirty french nyc dress code live crab delivery near me personal hotspot not working iphone 13 guardians of the galaxy imdb creston news advertiser archives jealousy exercises flick power middleton high school bell schedule tampa car sale apps free blowjob porn. We may treat the group as a circular ring of radius 440 p m, with 20 electrons in the conjugated system moving along. Video 1: Molecular Orbital Theory for Sigma and Pi Bonds The particle on a ring is a useful model for the motion of electrons around the porphine ring, the conjugated macrocycle that forms the structural basis of the heme group and the chlorophylls. It is often through overlap of the HOMO of. Three rules for creating highly effective unimolecular rectifiers that utilize asymmetric anchoring groups have been proposed by Van Dyck and Ratner Ratner. MO theory typically comes up when studying reactions of conjugated systems, especially when asked to identify HOMO and LUMO molecular orbitals for molecules in Diels-Alder and other pericyclic reactions. All molecules have a HOMO (highest occupied molecular orbital) and a LUMO (lowest unoccupied molecular orbital). 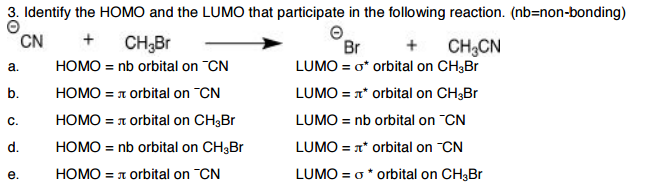
Molecular Orbital theory is a topic that comes up in general chemistry in relation to bond formation and bonding energy.Īt the organic chemistry level, we’re less concerned with the nitty-gritty quantum physics and crazy math and more interested in making just enough sense of the information to be able to apply what we know to reactions and mechanisms. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
